ADC Technology
How is AimedBio different?
ADCs have become a transformative approach in cancer therapy, offering a targeted means of delivering potent cytotoxic agents directly to cancer cells while sparing healthy tissue. By binding to specific tumor antigens, ADCs provide precision in treatment that traditional therapies like chemotherapy cannot.
Recent advancements have also underscored the importance of combination therapies, particularly with immune checkpoint inhibitors (ICIs). ICIs re-activate the immune system's ability to detect and fight cancer, but many tumors develop resistance mechanisms that reduce their efficacy. In response, pairing ICIs with ADCs has demonstrated potential to significantly improve clinical outcomes. ADCs can modulate the tumor microenvironment, enhancing immune system recognition and allowing ICIs to be more effective. This synergy was demonstrated in the combination of Pembrolizumab and Enfortumab Vedotin, setting a new standard for ICI-ADC combinations. As the demand for such combination therapies grows, the focus has shifted toward developing ADCs that are not only potent but also safe enough to be integrated into these regimens.
At AimedBio, we are committed to addressing this need by developing the next generation of safe and effective ADCs. One of our greatest strengths comes from our founder and clinical advisory board, who are actively engaged in patient care. This direct involvement allows us to maintain close collaboration with clinicians, gaining valuable insights into the most pressing medical challenges. In parallel, we conduct rigorous data analysis of patient-derived samples and public databases to identify the most promising and safe therapeutic targets. This dual approach—combining clinical connections and advanced data analytics—ensures that the ADCs we develop are both highly effective and precisely focused on areas where they can have the greatest impact.
Our integrated ADC development technology rests on three core pillars:
- Multi-omics platform: AimedBio stays updated on high-priority clinical needs through close collaboration with clinicians. By maintaining this collaboration, they ensure they are addressing the most pressing medical challenges. Additionally, through data analysis of patients' clinical data and public databases, AimedBio selects genes that are most effective targets for therapy. This dual approach ensures that the ADCs developed are both focused on the most impactful areas and highly effective.
- ADC platform: AimedBio’s proprietary PDC-based antibody panning method screens antibodies that can specifically recognize and bind to target conformations, enhancing the precision and effectiveness of the ADCs developed.
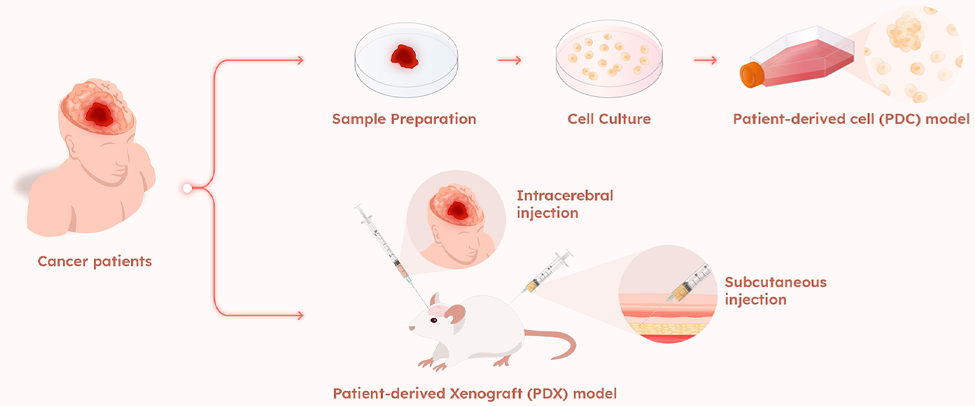
- Patient-derived model (PDC/PDX) platform: Utilizing Patient-derived cells(PDCs) allows AimedBio to reduce the high attrition rates often seen in clinical trials, providing a more reliable predictions of drug efficacy and safety. This approach saves time and resources by focusing on the most promising drug candidates, thereby accelerating the overall drug development process.
By maintaining all critical capabilities in-house, we are able to streamline every stage of the development process—from target discovery through clinical testing—ensuring that our innovative therapeutics reach patients faster and more efficiently.
 BBB Crossing Platform
BBB Crossing Platform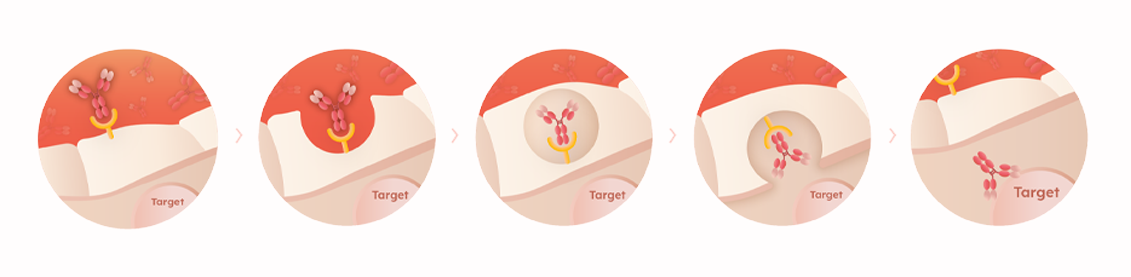
 Brain Immune Modulation Platform
Brain Immune Modulation Platform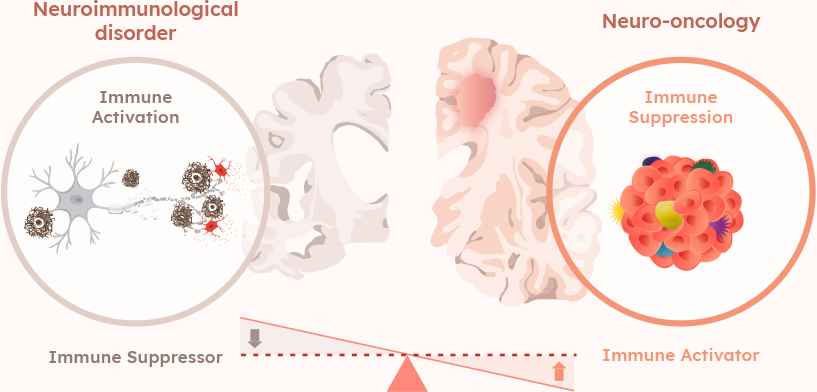
 Antibody-Drug Conjugate Platform
Antibody-Drug Conjugate Platform


 Multi-Omics Platform
Multi-Omics Platform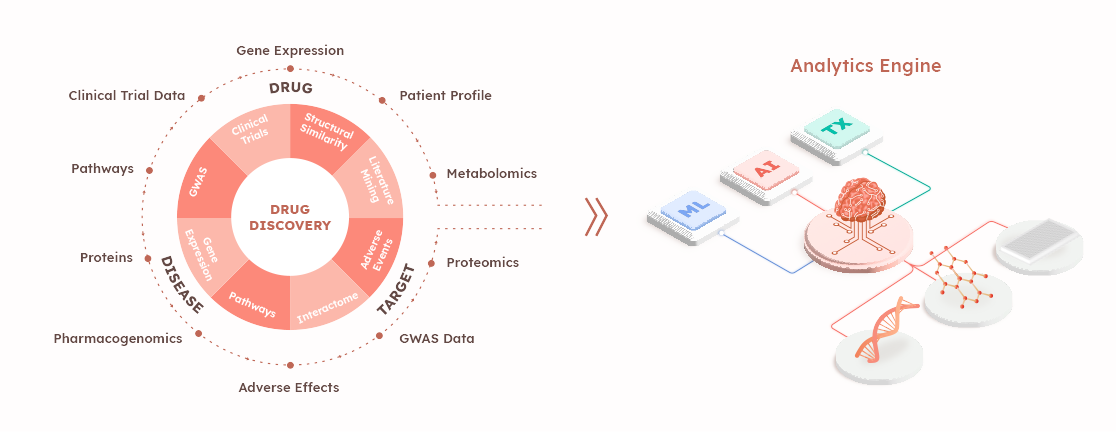
 Patient-derived model (PDC/PDX) Platform
Patient-derived model (PDC/PDX) Platform